Compliance
Navigate Complex
DSCSA Requirements with Confidence
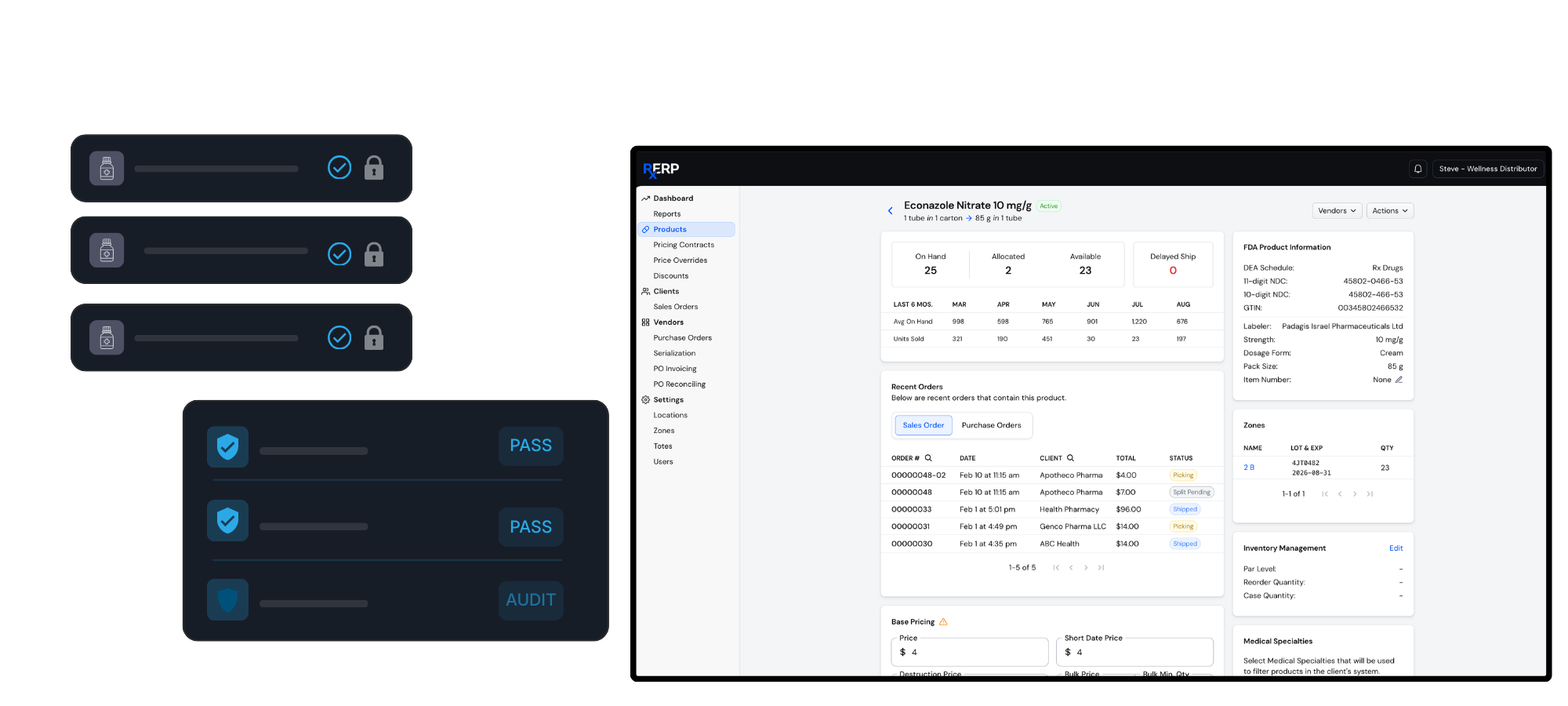
Our platform provides end-to-end compliance tools designed specifically for pharmaceutical operations, ensuring you meet DSCSA standards while protecting patient safety and supply chain integrity.
Key Serialization Features
Automated Serialization for DSCSA Compliance
Streamline your operations with full-scale supply chain management that ensures product integrity from manufacture to distribution. The RxERP platform provides end-to-end visibility across inventory management, product serialization, and quality assurance protocols that meet industry standards.
Comprehensive Reporting
Access detailed serialization reports for audits and regulatory submissions
EPCIS
Integration
Seamlessly manage Electronic Product Code Information Services data for supply chain partners
Unit-Level
Tracking
Automatically serialize each prescription medication to meet federal mandates
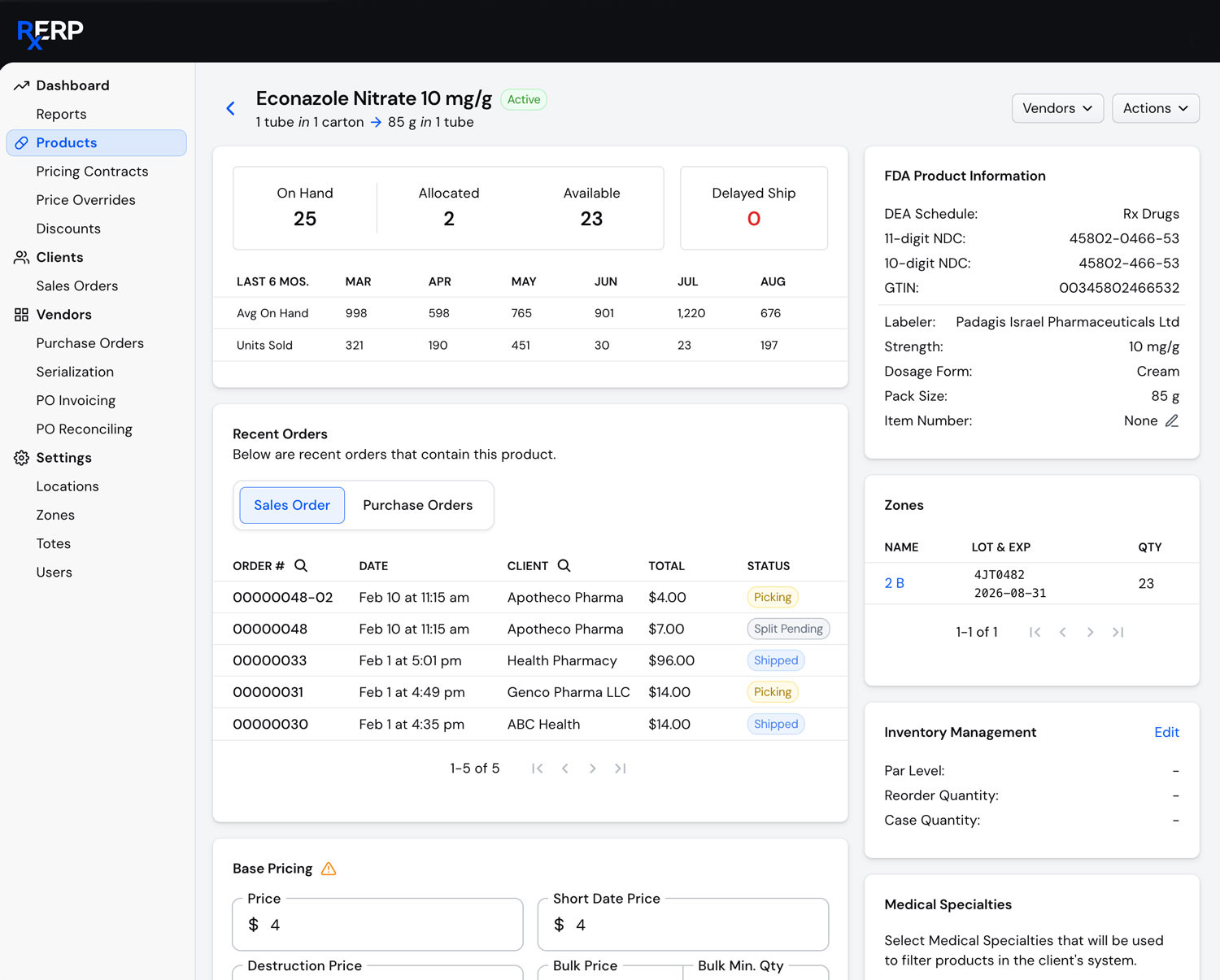
Counterfeit Prevention
Identify and prevent distribution of fraudulent medications through verification systems
T4 Report Generation
Streamline transaction history, transaction information, transaction statement and transaction tracking documentation
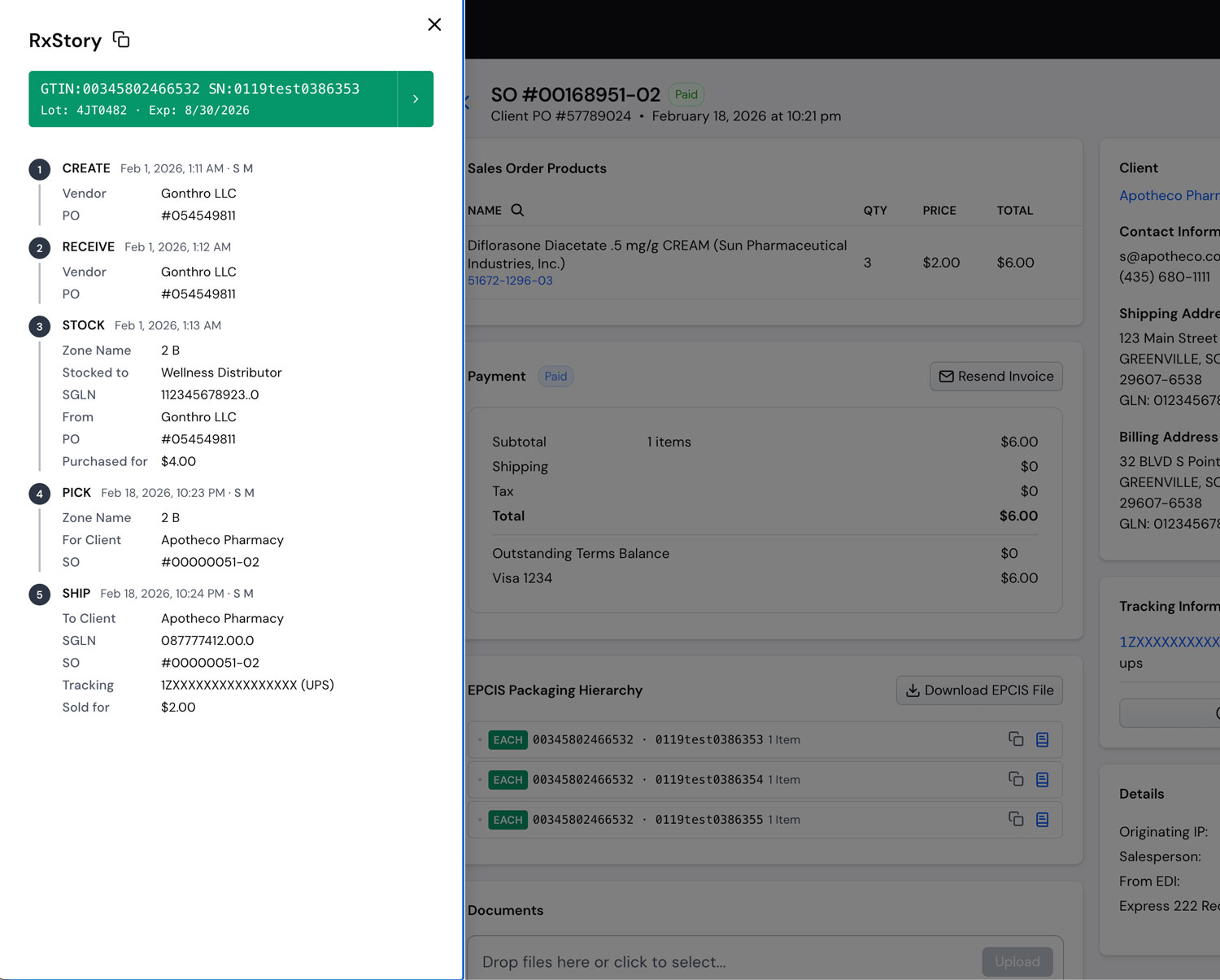
Traceability Benefits
End-to-End Traceability
Maintain complete visibility across your pharmaceutical supply chain. Our traceability solutions provide real-time tracking of products from origin to final destination, simplifying audits and enhancing operational transparency.
Complete Product
History
Track every transaction and movement throughout the supply chain
Simplified
Audits
Generate comprehensive reports instantly with historical data
Risk
Mitigation
Quickly identify and isolate affected products in recall scenarios
Enhanced
Transparency
Provide stakeholders with real-time visibility into product location and status
Regulatory
Documentation
Automatically maintain records built for FDA inspections and compliance reviews
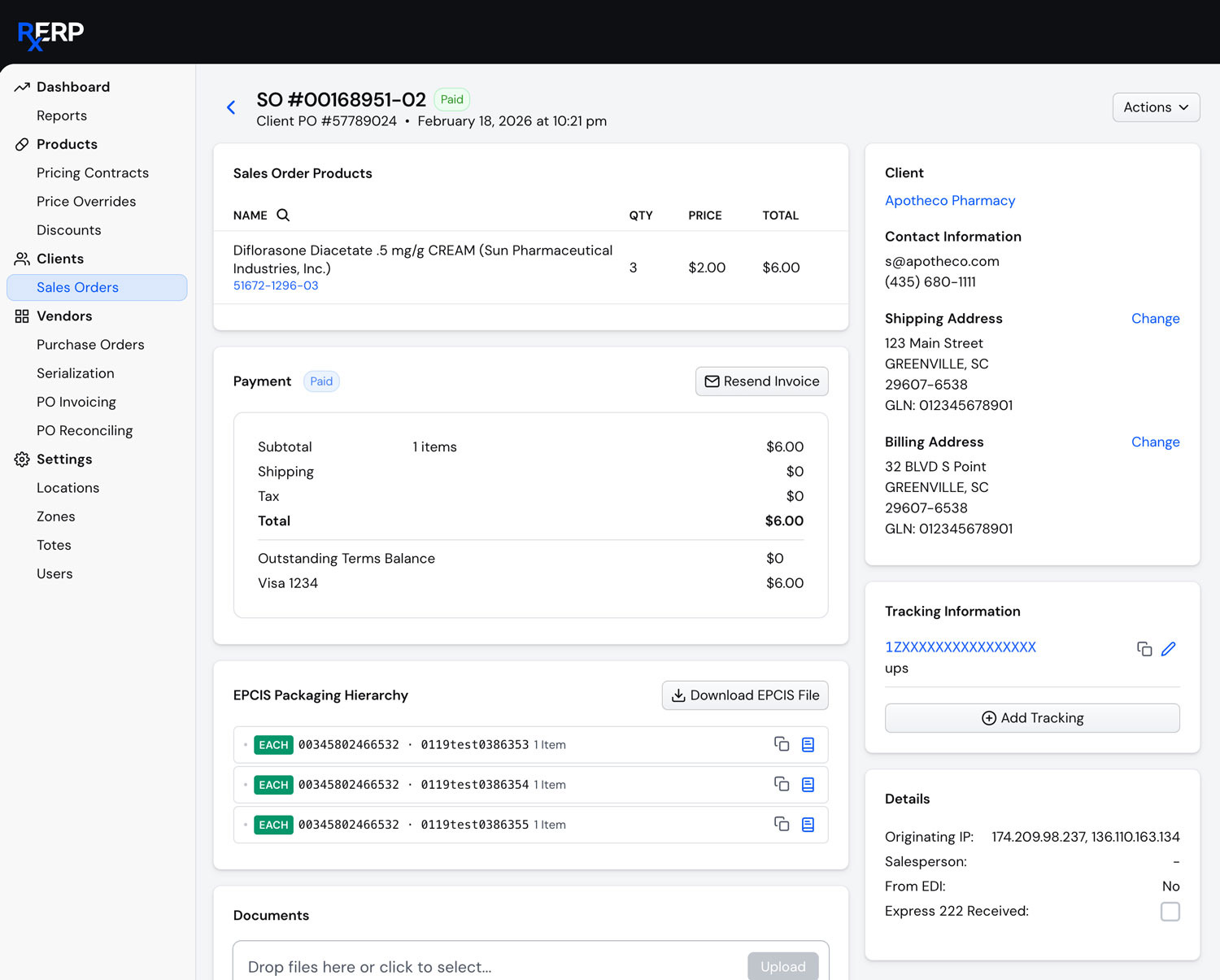
License Management Capabilities
Intelligent License Management
Ensure continuous compliance with automated license tracking and validation. Our system monitors all client and tenant licensure across jurisdictions, alerting you to upcoming expirations and regulatory changes.
Centralized
Tracking
Monitor all licenses, permits, and certifications in one platform
Automated
Validation
Verify license status and authenticity against regulatory databases
Expiration
Alerts
Receive proactive notifications before licenses expire
Multi-Tenant
Support
Manage compliance across multiple locations and business entities
Your Partner in DSCSA Readiness
RxERP simplifies pharmaceutical compliance through automation, integration, and intelligent workflows. Our platform is built by industry experts who understand the unique challenges of maintaining regulatory compliance while optimizing operational efficiency.
Whether you’re preparing for upcoming DSCSA requirements or enhancing existing compliance programs, our team is ready to help you assess your needs and implement the right solutions for your organization.
